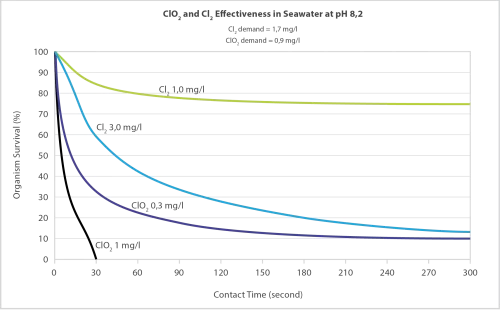
The oxidising properties and the radical nature of Chlorine Dioxide make it an excellent virucidal and bactericidal agent in a large pH range.
In alkaline media the permeability of living cell walls to gaseous chlorine dioxide radicals seems to be increased allowing an easier access to vital molecules.
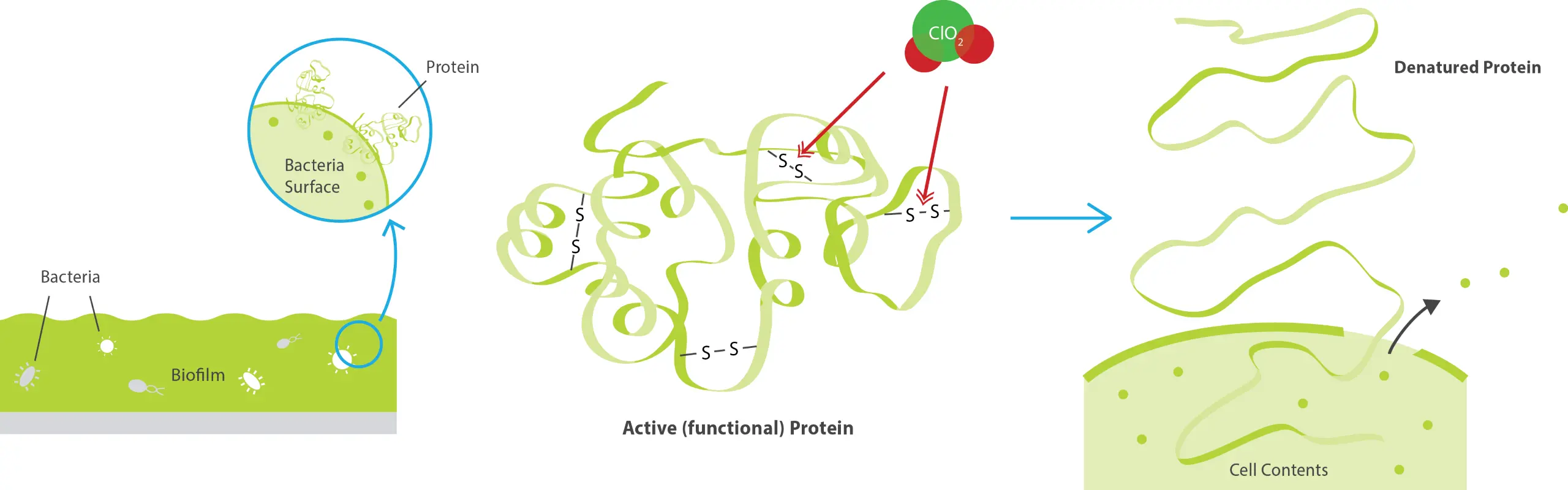
The reaction of chlorine dioxide with vital amino acids is one of the dominant processes of its action on bacteria and viruses.
Compounds within the cells and on the surface of cell membranes that contain oxidisable material react with chlorine dioxide, causing cell metabolism to be disrupted. Chlorine dioxide also reacts directly with disulphide bonds in the amino acids and the RNA in the cell.
Unlike non-oxidizing disinfectants, chlorine dioxide kills microorganisms even when they are inactive. The oxidative load placed on the cells by the action of chlorine dioxide mean that most microorganisms are unable to build up resistance to chlorine dioxide.